Mabs targeting programmed cell death 1 pd 1 programmed cell death ligand 1 pd l1 and cytotoxic t lymphocyte associated antigen 4 immune checkpoints have received regulatory approval across a wide range of tumor types including non small cell lung cancer nsclc.
First line therapy for non small cell lung cancer.
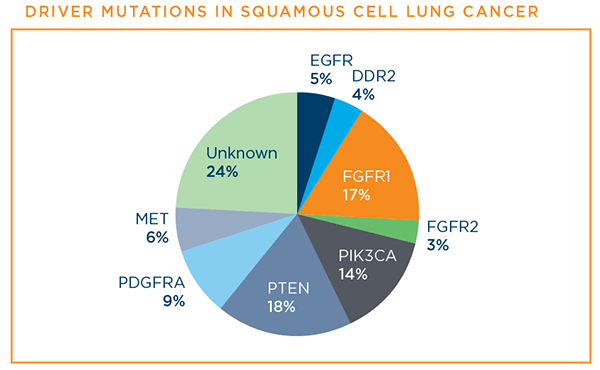
Necitumumab plus gemcitabine and cisplatin versus gemcitabine and cisplatin alone as first line therapy in patients with stage iv squamous non small cell lung cancer squire.
With the availability of several epidermal growth factor receptor egfr tyrosine kinase inhibitors tkis sequential therapy could potentially render egfr mutation positive non small cell lung cancer a chronic disease in some patients.
Among patients with a tumor proportion.
An open label randomised controlled phase 3 trial.
But other factors such as a person s overall health and lung function and if the cancer cells have certain gene or protein changes can also be important.
Medical treatment of advanced nonsmall cell lung cancer nsclc has been improved over the last two decades with the main increase in the number of active drugs the development of effective regimens and the introduction of salvage therapy after failure of first line treatment.
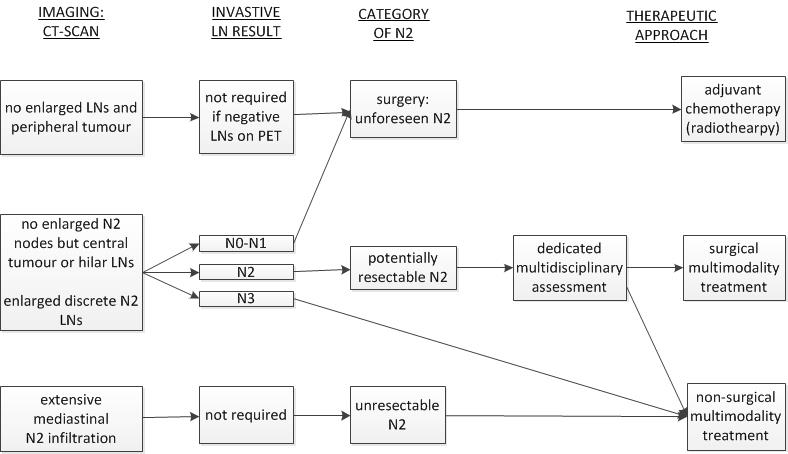
The treatment options for non small cell lung cancer nsclc are based largely on the stage extent of the cancer.
Immunotherapy has fundamentally changed the treatment landscape for many patients with cancer.
Options include erlotinib ramucirumab and nivolumab.
Third line chemotherapy is given for advanced or recurrent non small cell lung cancer nsclc after disease progression following first line and second line therapy.
Thatcher n hirsch fr luft av szczesna a ciuleanu te dediu m et al.